Row 3.
Group 1.
Analysis and Conclusions.
1. atomic radius: the average distance from the center of an atoms nucleus to its farthest out electron in the electron cloud.
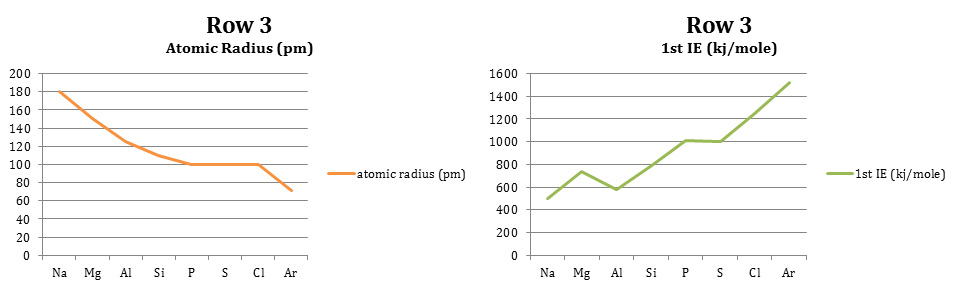
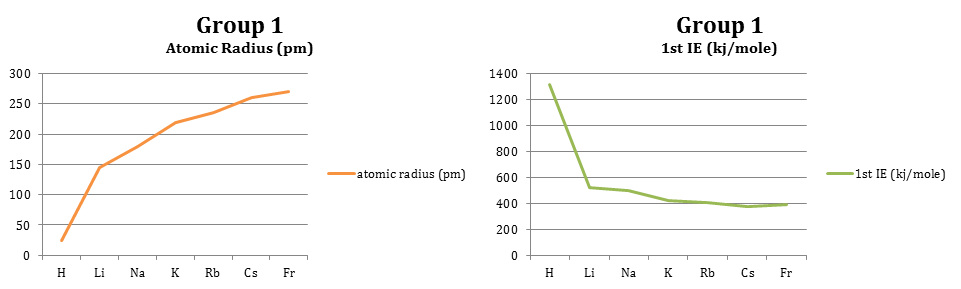
2. In row 3 as you move across the row it is noted that the average atomic radius decreases. This could be because as you move from left to right, there are more protons being gathered which pull more on the electron cloud, bringing the electrons in closer for a smaller radius. As you move down group 1, you can see that the average atomic radius increases most likely due to the addition of energy levels and electrons.
3. Factors determining trends in atomic radius are energy levels when moving up and down and proton numbers when moving left to right.
4. In ionic radii, when there is an addition of an electron, the atom gets larger because of the shielding effect and possible creation of another energy level. Also, the electrons repel each other in the negative to negative attraction. When an atom loses an electron, it could possibly lose an electron and an energy level but the electrons could also experience a greater pull from the positively charged nucleus.
5. first ionization energy: the amount of energy in kj/mole that it takes to remove the first electron.
6. In row 3 as you move across it is seen that the first ionization energy increases. This could be because as an atom becomes more like a noble gas, it does not want to lose electrons. When it is finally a noble gas (for example, Ar) it has 8 electrons and does not want to lose or gain one. As you move down group 1 the ionization decreases possibly because the atomic radius is increasing. If an atom has six energy levels, it is less likely to feel the effects of losing one or two electrons from the outer shell than an atom with one energy level losing electrons from the outer shell.
7. Factors determining the trends in first ionization energy include energy level size and number of electrons in relation to becoming a noble gas and having energy levels completely filled. The ionization energies are not smooth as you move across the row due to irregularities involving half filled and filled sublevels (s, p, d, and f) that repel paired electrons in orbitals. This repulsion also makes the electrons easier to remove.
8. Electron affinity increases up and to the right. First ionization energy also increases up and to the right. Electron affinity is adding an electron to an atom. Both electron affinity and ionization levels increase in the same way because it is going to be just as hard to add an electron as it will be to remove one from an atom that wants to have 8.
9. Characteristics of a metal include being on the left of the periodic table, good conductors of electricity and heat, strong, shiny, malleable, ductile, solid at room temperature, a high melting point, and usually form positive ions. Characteristics of a nonmetal include being on the right of the periodic table, brittle, does not conduct electricity or heat well, dull, gases at room temperature, and usually form negative ions. Metalloids are on the staircase inside of the periodic table. They usually look metallic but are brittle and are good semi-conductors. They do not conduct or insulate well. Metallioids are usually solids at room temperature. Noble gases, or group 18, are highly unreactive. They have 8 electrons and are all colorless and odorless gases at room temperature.
10. Row 3, or period 3, elements are classified as follows. Na (sodium) is a metal, Mg (magnesium) is a metal, Al (aluminum) is a metal, Si (silicon) is a metalloid, P (phosphorus) is a nonmetal, S (sulfur) is a nonmetal, Cl (chlorine) is a nonmetal, and Ar (argon) is a noble gas.
11. Group 1 elements are the most reactive metals because they have one electron in their outer shell. It is easy for these elements to lose one electron and become like a noble gas. Group 17 elements are the most reactive nonmetals because they have seven electrons in their outer shell. It is easy for these elements to gain one electron to become like a noble gas.
2. In row 3 as you move across the row it is noted that the average atomic radius decreases. This could be because as you move from left to right, there are more protons being gathered which pull more on the electron cloud, bringing the electrons in closer for a smaller radius. As you move down group 1, you can see that the average atomic radius increases most likely due to the addition of energy levels and electrons.
3. Factors determining trends in atomic radius are energy levels when moving up and down and proton numbers when moving left to right.
4. In ionic radii, when there is an addition of an electron, the atom gets larger because of the shielding effect and possible creation of another energy level. Also, the electrons repel each other in the negative to negative attraction. When an atom loses an electron, it could possibly lose an electron and an energy level but the electrons could also experience a greater pull from the positively charged nucleus.
5. first ionization energy: the amount of energy in kj/mole that it takes to remove the first electron.
6. In row 3 as you move across it is seen that the first ionization energy increases. This could be because as an atom becomes more like a noble gas, it does not want to lose electrons. When it is finally a noble gas (for example, Ar) it has 8 electrons and does not want to lose or gain one. As you move down group 1 the ionization decreases possibly because the atomic radius is increasing. If an atom has six energy levels, it is less likely to feel the effects of losing one or two electrons from the outer shell than an atom with one energy level losing electrons from the outer shell.
7. Factors determining the trends in first ionization energy include energy level size and number of electrons in relation to becoming a noble gas and having energy levels completely filled. The ionization energies are not smooth as you move across the row due to irregularities involving half filled and filled sublevels (s, p, d, and f) that repel paired electrons in orbitals. This repulsion also makes the electrons easier to remove.
8. Electron affinity increases up and to the right. First ionization energy also increases up and to the right. Electron affinity is adding an electron to an atom. Both electron affinity and ionization levels increase in the same way because it is going to be just as hard to add an electron as it will be to remove one from an atom that wants to have 8.
9. Characteristics of a metal include being on the left of the periodic table, good conductors of electricity and heat, strong, shiny, malleable, ductile, solid at room temperature, a high melting point, and usually form positive ions. Characteristics of a nonmetal include being on the right of the periodic table, brittle, does not conduct electricity or heat well, dull, gases at room temperature, and usually form negative ions. Metalloids are on the staircase inside of the periodic table. They usually look metallic but are brittle and are good semi-conductors. They do not conduct or insulate well. Metallioids are usually solids at room temperature. Noble gases, or group 18, are highly unreactive. They have 8 electrons and are all colorless and odorless gases at room temperature.
10. Row 3, or period 3, elements are classified as follows. Na (sodium) is a metal, Mg (magnesium) is a metal, Al (aluminum) is a metal, Si (silicon) is a metalloid, P (phosphorus) is a nonmetal, S (sulfur) is a nonmetal, Cl (chlorine) is a nonmetal, and Ar (argon) is a noble gas.
11. Group 1 elements are the most reactive metals because they have one electron in their outer shell. It is easy for these elements to lose one electron and become like a noble gas. Group 17 elements are the most reactive nonmetals because they have seven electrons in their outer shell. It is easy for these elements to gain one electron to become like a noble gas.
Application of Ideas.
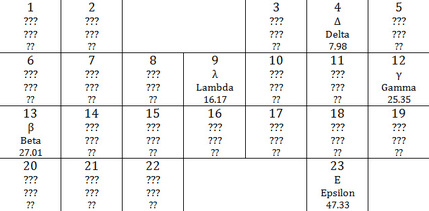
1. Create a periodic table:
2. The atomic number of an element with an atomic mass of 11.29 with nonmetallic properties and that is very reactive would probably be 6.
3. The atomic number of an element with an atomic mass of 15.02 that exhibits metallic properties but is softer than lambda and harder than beta would probably be 8.
4. The trends on my periodic table are alike the real periodic table in many ways because mine was modeled off of it. Atomic radius increases down and to the left while first ionization energy and electron affinity increase up and to the right.
5. The element with the largest atomic radius would be the element with the atomic number of 20. The element with the smallest atomic radius would be atomic number 5. The element with the highest first ionization energy would be atomic number 5. The element with the lowest first ionization energy would be atomic number 20.
3. The atomic number of an element with an atomic mass of 15.02 that exhibits metallic properties but is softer than lambda and harder than beta would probably be 8.
4. The trends on my periodic table are alike the real periodic table in many ways because mine was modeled off of it. Atomic radius increases down and to the left while first ionization energy and electron affinity increase up and to the right.
5. The element with the largest atomic radius would be the element with the atomic number of 20. The element with the smallest atomic radius would be atomic number 5. The element with the highest first ionization energy would be atomic number 5. The element with the lowest first ionization energy would be atomic number 20.