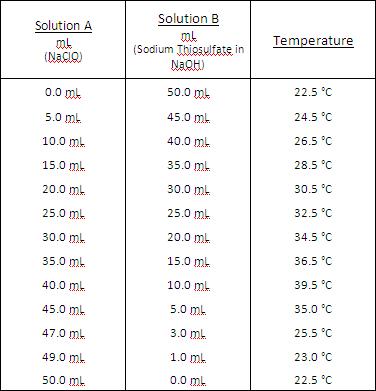
Data Table.
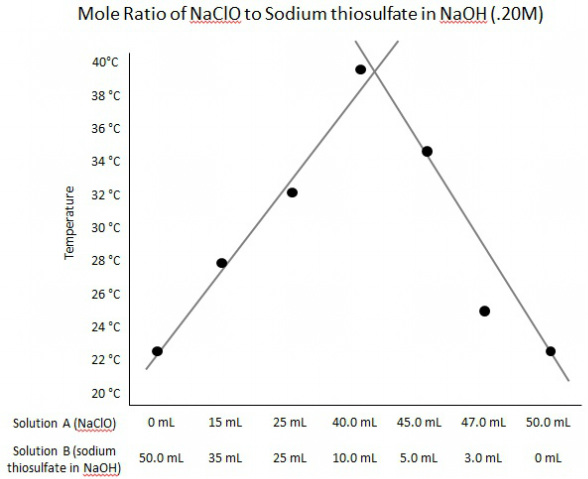
Graphs.
Conclusion.
In conclusion, the mole ratio of 40mL Solution A to 10mL Solution B was the optimum mole ratio for this reaction. In the experiment, the different mole ratios were tested to see which would provide the greatest reaction. Many mole ratios had to be tested between the two solutions to see which would give the highest yield in the reaction. Solution A and B were both tested at a mole ratio of 50 mL to 0 mL which provided the beginning temperature to compare to, 22.5°C. The peak temperature was found with 40mL of Solution A and 10mL to Solution B at a temperature of 39.5°C.
Discussion of Theory.
This method allows the mole ratio of reactants to be found by creating a 'guessing game' of trying to find the right ratios. The amount of reactants is changed each time to try to find the optimum quantity. In this case the optimum quantity was 40mL of Solution A and 10mL of Solution B. Since the highest temperature was recorded at 40mL of Solution A and 10mL of Solution B and the next recording taken at 45mL Solution A and 5mL of Solution B was lower, the assumption can be made that the optimum mole ratio is around 40mL of Solution A and 10mL of Solution B. This means Solution A has a 4 mole to 1 mole ratio with Solution B.
Analysis Questions.
1. A constant volume of reactants must be kept in order to be sure that each part of the experiment is carried out in the same way. For example, if 10mL of Solution A is used and there can only be a maximum of 50mL of the two solutions and somehow 42mL of Solution B is used, the reaction will be inaccurate.
2. "Limit reagent" means the reactant that limits the reaction or the reactant that is completely used up in the reaction.
3. Volume limits the precision of the data due to the fact that only a certain amount can be used as a total. The beakers used were no larger than 50mL so the absolute maximum amount of reactants that could have been used was 50mL. The beakers and graduated cylinders used were not absolutely perfect and most likely still had left over solutions in them, even after cleaning. Temperature did not limit the precision of the data because the same thermometer was used in each trial.
4. Solution A is the limiting reagent along the upward slope of the line and Solution B is the limit reagent along the downward slope of the line.
5. Other physical properties other than temperature change that could use the method of continuous variation are color change and odor. Both can become stronger as the chemical reaction gets stronger.
6. It is more accurate to use the point of intersection of the two lines to find the mole ratio rather than the ratio associated with the greatest temperature change because it might not be the best mole ratio. For example, if the ratio with the greatest temperature change is 40mL of Solution A to 10mL of Solution B, and no other ratios are tested within the 5mL changes in the other parts of the experiment, there is no way of knowing if 42mL of Solution A and 8mL of Solution B will be the better mole ratio for the temperature change. There is no way of knowing until the mole ratio is tested.
2. "Limit reagent" means the reactant that limits the reaction or the reactant that is completely used up in the reaction.
3. Volume limits the precision of the data due to the fact that only a certain amount can be used as a total. The beakers used were no larger than 50mL so the absolute maximum amount of reactants that could have been used was 50mL. The beakers and graduated cylinders used were not absolutely perfect and most likely still had left over solutions in them, even after cleaning. Temperature did not limit the precision of the data because the same thermometer was used in each trial.
4. Solution A is the limiting reagent along the upward slope of the line and Solution B is the limit reagent along the downward slope of the line.
5. Other physical properties other than temperature change that could use the method of continuous variation are color change and odor. Both can become stronger as the chemical reaction gets stronger.
6. It is more accurate to use the point of intersection of the two lines to find the mole ratio rather than the ratio associated with the greatest temperature change because it might not be the best mole ratio. For example, if the ratio with the greatest temperature change is 40mL of Solution A to 10mL of Solution B, and no other ratios are tested within the 5mL changes in the other parts of the experiment, there is no way of knowing if 42mL of Solution A and 8mL of Solution B will be the better mole ratio for the temperature change. There is no way of knowing until the mole ratio is tested.