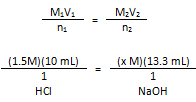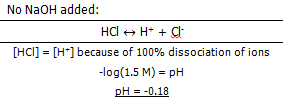Purpose.
To find how much of NaOH it takes to properly titrate and neutralize 10.0 mL of 1.5 M HCl.
Procedure.
1. Measure 10.0 mL of 1.5 HCl into a graduated cylinder and pour into a 50.0 mL Erlenmeyer flask.
2. Add two drops of phenolphthalein into the beaker of HCl. The color will remain clear but when the NaOH is added, it will turn pink to show the neutralization.
3. Record your initial amount of NaOH in a data table.
4. Add NaOH until the color starts to change. Swirl the flask until the pink color is hard to swirl away. Keep adding NaOH until the solution is a light pink. Record the changed amount of NaOH.
5. Dispose of the neutralized solution, rinse out the Erlenmeyer flash and repeat twice. Refill the NaOH if needed.
2. Add two drops of phenolphthalein into the beaker of HCl. The color will remain clear but when the NaOH is added, it will turn pink to show the neutralization.
3. Record your initial amount of NaOH in a data table.
4. Add NaOH until the color starts to change. Swirl the flask until the pink color is hard to swirl away. Keep adding NaOH until the solution is a light pink. Record the changed amount of NaOH.
5. Dispose of the neutralized solution, rinse out the Erlenmeyer flash and repeat twice. Refill the NaOH if needed.
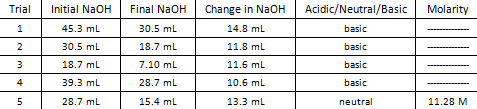
Data Table.
Calculations.
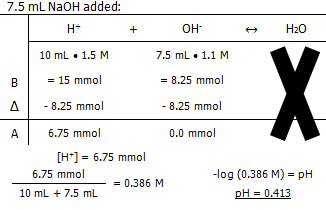
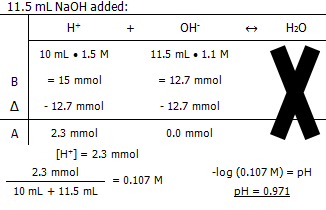
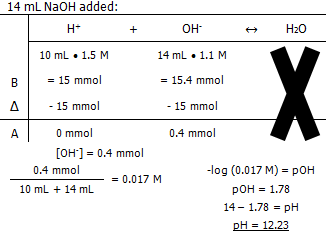
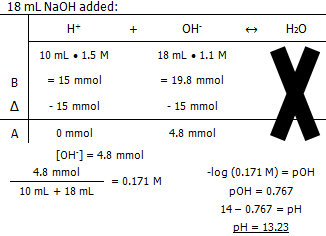
Molarity of the NaOH
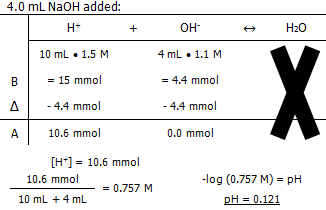
The pH of the solution after:
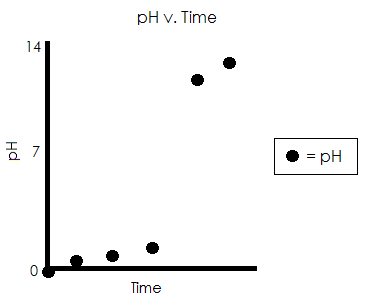
Graph of pH v. Time for Above Calculations
Conclusion.
In conclusion, it takes 13.3 mL of 1.1 M NaOH to titrate 10 mL of 1.5 M HCl.
Discussion of Theory.
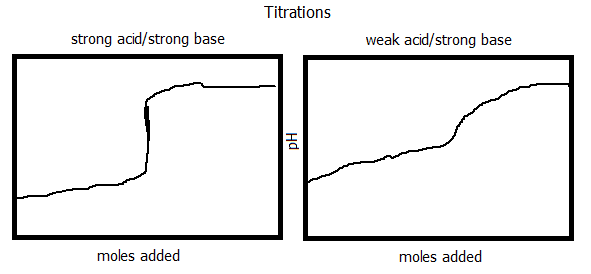
Buffered solutions are solutions that resist a change in pH when either H+ or OH- ions are added. It prolongs the change in pH. Buffered solutions contain either a weak acid or a weak base and it's salt (conjugate). In all acid-conjugate base pairs, all solutions will have the ratio [A-]/[HA] will have the same pH where [A-] is the conjugate base and [HA] is the acid. When this is true, the Henderson-Hasselbach equation can be used. In a buffered solution, there is a buffer capacity. This is the amount of H+ or OH- ions that the buffer can absorb without significantly changing the pH. In a titration, there is a controlled addition of a solution of a known concentration in order to determine the unknown concentration of a solution. There is an equivalence point that is reached when the reaction between the titrant and the unknown has been completed. The equivalence point can be shown by an indicator, such as the phenolphthaline used in this experiment. The indicator changes the color of the reaction once the optimum pH (equivalence point) has been reached. This color change is also known as the end point of a titration. Most indicators begin as a weak acid and shift to their conjugate base, causing the color change. Each indicator must be carefully chosen to fit with the correct equivalence pH change in the titration. In a strong acid/strong base titration, prior to reaching the equivalence point, the pH can easily be calculated by using the [H+]. The equivalence point will always have a pH of 7.00. Past reaching the equivalence point, the [OH-] can be calculated from excess OH- and the volume of the solution. The [H+] and pH can then be calculated. In a titration between a weak acid and a strong base, the reaction must first be assumed to run to completion. In such reactions, the concentration of the acid remaining and the conjugate base must be found. Once these are found, the calculation is a simple equilibrium problem to calculate pH. The pH at the equivalence point of a weak acid and strong base titration is always greater than 7.00. When titrating a weak base with a strong acid, the pH will always be below 7.00 at the equivalence point. In each titration, the major species must be decided. Then it must be decided if the equation will react to completion. If so, the equivalence point may be found and ultimately the pH.
Questions.
1. The purpose of doing a titration is to neutralize a solution. In this case, the purpose of the titration was to find the molarity of the NaOH added.
2. An indicator is used to determine the equivalence point of a titration. Most are weak acids that shift from acid to conjugate base forms. The color change of the indicator occurs when a proton is lost or gained and when the concentration of the more dominant form is ten times as great as the less dominant form.
3. To determine what indicator to use, find one based on the equivalence point. Each indicator has a different pH where it will change color. One must be found and it's color change point must be matched with the pH where the solution will be neutralized in the titration process.
4. The equivalence point is the point in the titration where the reaction between the titrant and unknown compound is completed. The equivalence point is the vertical line on a titration curve. The end point is the point in the reaction where the indicator turns a color. The indicator may also change color at the equivalence point.
5. Compare and sketch a titration graph for a strong acid/strong base titration and a weak acid/strong base titration.
2. An indicator is used to determine the equivalence point of a titration. Most are weak acids that shift from acid to conjugate base forms. The color change of the indicator occurs when a proton is lost or gained and when the concentration of the more dominant form is ten times as great as the less dominant form.
3. To determine what indicator to use, find one based on the equivalence point. Each indicator has a different pH where it will change color. One must be found and it's color change point must be matched with the pH where the solution will be neutralized in the titration process.
4. The equivalence point is the point in the titration where the reaction between the titrant and unknown compound is completed. The equivalence point is the vertical line on a titration curve. The end point is the point in the reaction where the indicator turns a color. The indicator may also change color at the equivalence point.
5. Compare and sketch a titration graph for a strong acid/strong base titration and a weak acid/strong base titration.
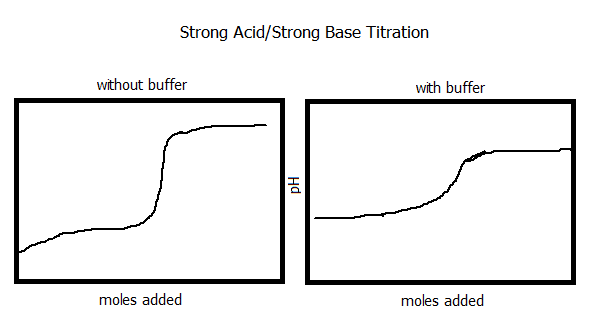
6. Compare and sketch a titration graph for a strong acid/strong base titration and the same titration after a buffer solution has been added.
7. A buffer is a weak acid or base and it's salt (conjugate). For example, HF and NaF are buffers. A buffer solution resists a change in pH when H+ or OH- ions are added. A buffer solution will prolong a reaction if added. When H+ or OH- ions are added to a solution, the weak acid or base is the best source of other H+ or OH- ions. The ions form to make water (H2O). The remaining conjugate reacts with more H+ or OH- to make a compound. If the concentration of the compound and it's conjugate are large compared to the concentration of H+ or OH- ions, there will be very little change in the pH.
8. a. H c. N e. H g. H
b. L d. N f. L
8. a. H c. N e. H g. H
b. L d. N f. L